-
Posts
38 -
Joined
-
Last visited
About iwannagofishin
- Birthday 10/31/1950
Personal Information
-
Real Name
Al
-
Biography
Ex commercial salmon troller, now a catch & release advocate.
-
Location
Courtenay, British Columbia, Canada
-
Interests
fishin 'n' huntin
-
Occupation
Fishing lure manufacturer
Recent Profile Visitors
The recent visitors block is disabled and is not being shown to other users.
iwannagofishin's Achievements
10
Reputation
-

Do you catch fish close to your boat?
iwannagofishin replied to iwannagofishin's topic in Salmon Pro's Connection
-

Do you catch fish close to your boat?
iwannagofishin replied to iwannagofishin's topic in Salmon Pro's Connection
-

Do you catch fish close to your boat?
iwannagofishin replied to iwannagofishin's topic in Salmon Pro's Connection
Hi Rossport. Actually, I have just moved back to Nipigon area (Red Rock for now). Your boat sounds fishy, especially if you are catching them close to the boat. Do you know if the hull is bare aluminum and have you tested for electrical continuity between the outboard anodes and the hull? If you have a kicker, check it as well. My first thought is that the mineral content of Superior might be low where you are right now and the attraction effect of the boat's signature might only reach so far. I assume that you are fishing the salmon lures deeper and further away from the boat than the 10ft mentioned. If so, are you catching lakers on the deeper lures, or just the ones closer to the boat? A 'rule of thumb' is that fish will normally strike the lure that is closest to the voltage source. In your case the voltage source will be the boat. A good way to test this theory is to place a voltage source closer to the deeper lures. This might be in the form of voltage tuned spoons, or a portable black box. These are both available at Lurecharge.ca . I sold this business a couple of years ago and have no monetary connection to their sales. I did however just publish a book. It is called Why Can't I Catch Fish With My Boat. It is available at Placertools.com in print, or in digital form on amazon. My email is included in the book and free online support is included. -
Hi again fellow anglers. A HAPPY NEW YEAR to you all. I've been away from the forum for awhile. Some of you may remember my posts and conversations about voltage tuned boats and voltage tuned lures. I recently sold my voltage tuned lure business, Lurecharge, to one of my customers. I was asked to stay on with the business as a technical advisor. Unfortunately, I lost my old email in the transition because of a website change, but was suppied with a new one, so responses to my posts should work again. This is an absolutely free service that I offer in order to help out anglers with their non-fishy boats. When I was marketing my voltage tuned lures, I came to the conclusion that my lures didn't work for everyone. For the ones that it did work for, they could generally catch 80% of the fish on one side of the boat if they only used one of my lures. This discovery led me to work on finding out why one boat would catch fish while a seemingly identical boat would not. This started years ago when I was commercial trolling for salmon on Canada's west coast. If your boat wasn't right, you didn't catch as much as the other guys. So, a few years ago, I placed adds on Kijiji 'Free Help for Anglers with Non-Fishy Boats'. Over a 2 year period, I was approached by many anglers and the results were such that I had a good cross section of varying scenarios. It was simply a matter of compiling the data and connecting the dots. Then it was up to me to personally test and prove the findings. This culminated with a 5 lake study over several months. Instead of using voltage tuned lures, I used identical plain ones and modified the signature of my 14' aluminum boat to be positive or negative. I was basically surface trolling for trout with one lure 30' behind the boat and a second, identical lure trolled 100' plus behind the boat. The results were such that I could make the 30' lure catch 3/4 of the fish, or the 100' lure catch 3/4 of the fish depending on how the signature of the boat was configured. When my boat had a positive voltage signature, it would attract fish to the 30' lure. When my boat had a more negative voltage signature, the fish were somewhat repelled by the boat. A good positive voltage signature requires a boat with bare underwater hull metals (cathode) that has an area that is at least 5 times the surface area of the anodes (hull ratio). There is a big range of ratios that seem to work well, from 5:1 up to 300:1. I found that about 100:1 worked best. Originally, my aluminum lake boat only had the small 8hp outboard zinc and my ratio was about 1000:1. The boat always fished ok, but I never had fish jump as close to the outboard as with 100:1 ratio. A bare aluminum boat without a connected outboard zinc will not catch fish very well either. I proved this one time while product testing my tuned lures one fall. There was a guy with a 12' cartopper and an electric trolling motor (no zinc). He fished all day with a lake troll with spinner and worm. At the end of the day, he had zero strikes while I recorded 17. This convinced that a bare aluminum boat will repel fish just like a boat that has a poor ratio, or simply too much zinc ratio (overzinced). These principles apply to all types of boats and most of them can either be fixed, or the negative impacts reduced to where voltage tuned lures will work very well. I have helped guys that their boat had such a strong negative signature that my voltage tuned lures did not help. These effects influence fish at greater depths that even surprised me. Not once in all my years has a customer ever said to me that they can only catch fish past a certain depth. It seems that a boat that can't catch fish, also can't catch fish at over 200'. This applies to both fresh and salt water. Here is a link to an article that I wrote in 2020. https://islandfishermanmagazine.com/is-your-boat-voltage-tuned-for-fishing/
-

Lund Tyee vs Lund Fisherman - Advice Needed
iwannagofishin replied to TrueNorth33's topic in General Discussion
I have been tuning non-fishy boats for a long time and I would suggest buying a boat that does not have a totally painted bottom. An aluminum boat that is totally painted and with a modern outboard that has too much anode surface area will actually repel fish. I have helped too many guys with that exact scenario to be wrong. Your catch ratio will go down by 2/3 and you will mark less fish on your sounder. I have sold my voltage tuned lure business (Lurecharge) to one of my customers, but am staying on as technical advisor. This is an absolutely free service. Al -
I help for free, just to build my database of problem boat issues. After years of research and testing, I have identified why one boat will catch fish and another won't. I did a 5 lake study over a couple of months, adjusting my boat's signature in water. To make a long story short, I coud effectively move the strike zone from 30 ft behind the boat to 100 ft behind the boat, simply by adjusting the setup between the boat metals and the anodes. I believe that about 80% of boats sold today fall far short of their potential. If you have bought a boat, and it doesn't catch fish as well as your last boat, or if you catch more fish on your back lines than on your front ones, send me an email, and I will help you to correct the issues. No fees, no obligations. [email protected]
-
Hi all. A while back, I promised some anglers that I would insert a link to an article that I wrote for the Island Fisherman Magazine. The article probably relates more to fresh water boats, simply because corrosion is not always thought of, yet the fish are just as affected in fresh as well as salt water. The magazine does not normally release articles so soon after publication, but the owner graciously agreed to let this one out for me. This may not happen again. The May issue will be - "DOWNRIGGER WIRE VOLTAGES and WHY WE HAVE BEEN GETTING IT WRONG ALL THESE YEARS". I will continue to submit articles that have at least a new twist on them. Any and all questions are welcomed. [email protected] - Be safe during this viral onslaught. https://islandfishermanmagazine.com/is-your-boat-voltage-tuned-for-fishing/
-

Does your boat fish 'good' or 'bad'?
iwannagofishin replied to iwannagofishin's topic in General Discussion
Once again, I make 'non-fishy ' boats catch fish. The help is free and the results are real. Just PM or email me [email protected] -

Does your boat fish 'good' or 'bad'?
iwannagofishin replied to iwannagofishin's topic in General Discussion
The most recent complaint came from a chap who had recently purchased an 18' Lund with a single outboard. He now claims to be at about a 30% catch rate of where he used to be with his old boat. I asked him some questions and found out that the boat was completely paint sealed at the factory. I made some suggestions on how to improve the fishability of his boat and am expecting some feedback soon. I will update on this topic when I hear from him. -

Do you catch fish close to your boat?
iwannagofishin replied to iwannagofishin's topic in Salmon Pro's Connection
Thanks for that, Jim. Funny that you say that about the old boat. I was explaining some of the concepts to a chap not long ago and he commented; Maybe that's why the old timers claimed that if you hung your catch over the side on one of those metal clip fish stringers that you would catch even more fish. I had not made that connection, but the ones I used back then were zinc coated. As long as they were in contact with the aluminum hull, you would change the hull's voltage signature, just like adding a small amount of zinc to the hull. Al -

Do you catch fish close to your boat?
iwannagofishin replied to iwannagofishin's topic in Salmon Pro's Connection
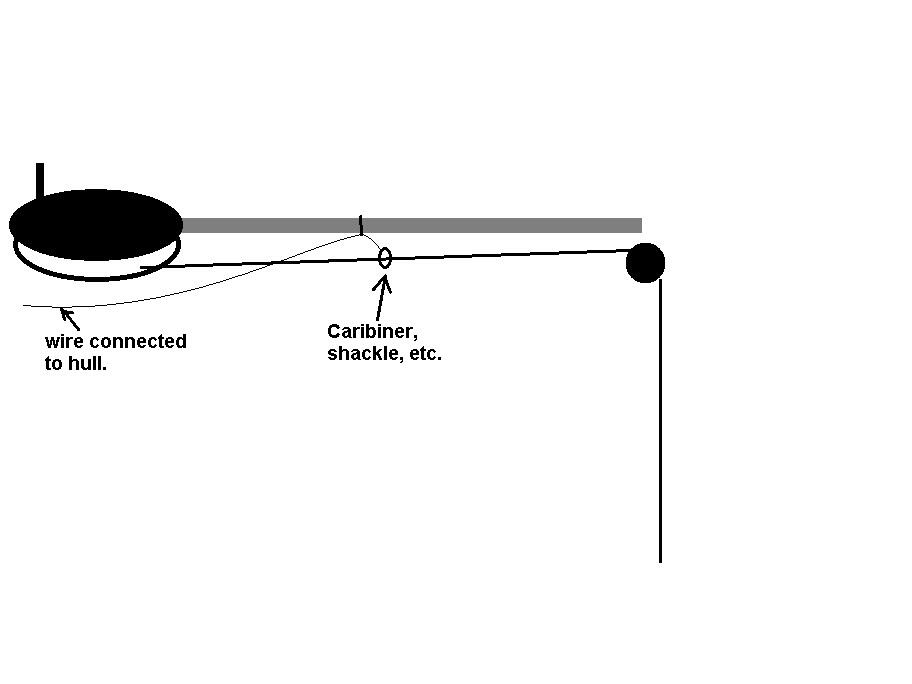
Thanks Superdon Yes to the transducer brackets, trim tabs, etc. In fact, any exposed, underwater stainless steel that is included in the bonding system will raise the overall hull voltage. This is simply because stainless will have a higher rating on the galvanic scale. I'm not saying that the fish like more, but It doesn't seem to hurt, at least to a point. Always listen to the fish. The 6 ft radius I am sure makes for a perfect distance of coverage. The loss of effect on metals that are further away will be quite minimal. An an example the one chap that I helped had a 20'aluminum boat. His cure was installing a jumper wire from the bottom of the leg to the steering bracket and having the continuity from his trim zinc through to the hull. He catches lots of fish now and asked if he could distribute some of my tackle in southern Ontario. He could not believe that a 3 ft piece of wire could put fish in the boat. So, his outboard zincs were, at least. trying to protect the entire boat. As far as the trolling wires, I have not seen much obvious repelling of fish with respect to stainless. The only 2 confirmed ones at this time are single metals of zinc and aluminum. An aluminum hull may fish ok without zincs, perhaps depending on the rivets or combination of metals. This will itself make a galvanic cell to some degree, depending on the galvanic rating of each of the combined metals. Like my lures, I use between 5 and 20% sacrificial metals to the rest being the proud metals with higher galvanic ratings. If you reverse the proportions, it may compromise the desired effect. You don't want the entire aluminum hull being the anode and protecting the rivets or other attached metals. Back to trolling wires. If they are isolated from the boat and bonding system, it is possible that they can be have a negative effect. Unless you are using ion control. Another option is to actually have the wires incorporated with the bonding system and giving off voltage. If you have ever tested wire voltage with a meter between the negative ground and the trolling wire, all you are reading is the potential. Once you remove the meter, no circuit exists. If you try this test, check the wire "potential", now drop the meter probe into the water beside the boat. You will find the readings to be almost the same. Think about that. The best way to check if you do have wire voltage is by how it is described in the diagram. This is different from the diagram that I included previously in this post. If someone has ion control, they can do this test with power on as well as power off, to confirm. The issue there will be that anytime that you have or apply voltage to something, it will act like a capacitor for up to 15 minutes while the voltage dissipates. You can watch the voltage come down on the meter as it dissipates and is usually gone in a couple of minutes. I also included a diagram on one option of including trolling wire with the bonding system. One mention. Some commercial trolling wire has brass markers (stoppers) every 9 to 15 feet. The brass markers would turn reddish color when the voltage was higher. They became the sacrificial anodes for the stainless. -

Do you catch fish close to your boat?
iwannagofishin replied to iwannagofishin's topic in Salmon Pro's Connection
I trolled commercially for many years and my biggest boat was a 54 ft freezer troller. I always used a Russell black box, which does the same essentially as does Cannon ion control. The fishiest boats seemed to be the steel ones, or ones with the most underwater metal. This is only true if they used copper bottom paint which still allows the water to interact with the steel. Aluminum boats cannot use copper paint unless they seal the aluminum completely as copper and aluminum make a great corrosion cell. The point here is that the more exposed underwater metal that you have that is bonded together and has the proper proportion of zincs or anodes will have a better chance of giving off a stronger positive (+) signature. If you have the same scenario on a boat where there is no bonding, or no zincs, it will give off a weaker signal or a negative (-) signature and repel fish. Some aluminum boats that have no zincs will repel some fish, guaranteed! I have also proven that a zinc alloy anode that is not in contact with and protecting a more proud metal, will repel some fish. I am talking about my own Lurecharge, salt water anodes here. They fit the Jekyll and Hyde scenario, and as long as they are in contact with a more proud metal, like stainless steel or nickel spoon, then a positive voltage will radiate from that spoon. So, if you have a boat with an inboard and ion control, it is still possible to repel fish from any non-bonded, non-zinced, exposed, underwater metals, even though there is a positive voltage coming from the ion controlled downrigger wires. If the boat is giving off a negative, it may be strong enough to overcome the ion control, even when you are fishing deeper. It is possible to have several signatures coming from a boat at the same time, unless you take care of the bonding and anodes. Fresh water is the worst because you simply don't see corrosion like you do in salt water, so guys think that they don't need to do anything. Voltages still exist and fish are still sensitive to it. -

Do you catch fish close to your boat?
iwannagofishin replied to iwannagofishin's topic in Salmon Pro's Connection
It does apply to fiberglass boats as well. Please look back 4 posts and I refer to a chap with a glass boat who had troubles catching fish. His basic problem is that he had too much underwater metals that were not connected to the bonding system (negative battery ground??). Every not-bonded metal will have it's own signature and some of these can be negative. This can ruin your fishing even though you have an acceptable reading from your engine and/or incomplete bonding system. Trim tabs, through hull fittings, ladders, metal bow protectors, props, trim anodes, etc, should all be interconnected to the bonding system to create only one voltage signature coming from the boat. Saying that, it sounds like your boat isn't broke and doesn't need fixing, as far as fishing goes. To test, complete all bonding and test as indicated in the attached diagram. If the engine has electric start, you can attach the negative meter probe to the negative ground or engine block. In fresh water, you should expect to see a positive voltage between .2 and .6 volts. This will vary greatly, depending on the mineral content of the water and to some degree, temperature. The use of a stainless reader wire on the positive end is not perfect for all situations, but gives the numbers that we are accustomed to seeing. A boat with a stainless prop or trim tabs will give higher readings than a boat with an aluminum prop, painted or not. -

Do you catch fish close to your boat?
iwannagofishin replied to iwannagofishin's topic in Salmon Pro's Connection
Keep in mind that the anodes are half of the cell and the spoon metals are the second half. If you use any copper or brass components or swivels, the voltage will be reduced. If you use tinned or zinc coated hooks, the effect will be lost. I prefer nickel or stainless components and spoon bodies that are not clear coated. Saying that, everything can be found at the Lurecharge website. -
Try the 4 Penny knot as shown in this video.